Article: The Physics & Chemistry of Stress - NuSpecianism SDAE Solution
The Physics & Chemistry of Stress - NuSpecianism SDAE Solution
Stress Chemistry & Stress Physics
How stress hormones reshape metabolism—and how that reshaping can contribute to chronic disease over time
Educational content only. Not medical advice. Stress biology varies by person and condition. If you have symptoms, diagnoses, or medication questions, work with a licensed clinician.
Why this matters in NuSpecies and SDAE
I’ve seen something clinically “human-true” in my consultations: when people understand how the body actually works, they often become more motivated and consistent—because knowledge turns vague fear into concrete action.
SDAE frames that motivation as a restoration of Inner–Outer coherence:
- Outer Reality (stressors, environment, systems, social injury) applies pressure.
- Inner Reality (meaning, attention, spirit, identity) determines whether that pressure becomes chronic curvature—physiologically and psychologically.
What follows is a science-grounded account of the mechanisms: how stress hormones change metabolism and how that can, over time, contribute to chronic disease risk and progression.
1) The two master stress systems: fast and slow
Most “stress biology” runs through two coupled systems:
The fast system: Sympathetic nervous system (SNS)
This triggers the release of catecholamines (primarily epinephrine and norepinephrine), quickly shifting heart rate, blood pressure, and fuel availability.
The slower system: Hypothalamic–pituitary–adrenal axis (HPA)
This drives secretion of cortisol (a glucocorticoid), which changes gene expression and metabolism over minutes to hours and follows a strong circadian pattern that can become disrupted under chronic stress.
In healthy cycles, these systems spike when needed and then resolve. Under chronic stress, they can become dysregulated—contributing to what stress researchers call cumulative physiological “wear and tear.”
2) Stress physics: the body reallocates energy under threat
A useful “physics lens” is to treat stress as energy re-budgeting.
Under threat, the body re-prioritizes:
- immediate ATP availability
- rapid glucose release
- rapid lipid mobilization
- heightened cardiovascular delivery
- immune system “mode switching” (short-term mobilization vs long-term dysregulation)
A mitochondria-centered review describes stress as an energy-dependent coordinated process, emphasizing how stress mediators influence energy substrate distribution and mitochondrial function.
SDAE translation: stress is not merely emotion—it is a systems-level reallocation of fuel, signaling, and repair capacity.
3) Stress chemistry: how catecholamines and cortisol “rewrite” metabolic pathways
3.1 Catecholamines: rapid signaling via receptors and second messengers
Catecholamines act through adrenergic receptors. A key pathway is β-adrenergic receptor → cAMP → protein kinase A (PKA) signaling, which rapidly changes enzyme activity.
One high-impact metabolic consequence: lipolysis (release of fatty acids and glycerol from fat tissue). Sympathetic outflow directly stimulates adipocyte lipolysis via β-adrenergic receptors and cAMP-dependent pathways.
This is adaptive short-term: you mobilize fuel fast.
3.2 Cortisol: slower genomic control (gene expression)
Cortisol binds the glucocorticoid receptor (GR), which functions as a transcriptional regulator—turning metabolic genes up or down over time.
In the liver, glucocorticoids stimulate gluconeogenesis by upregulating enzymes such as PEPCK and glucose-6-phosphatase (G6Pase).
Cortisol also shifts lipid and protein metabolism in ways that are helpful during acute stress but can become harmful when persistent. Reviews link excess or prolonged glucocorticoid signaling with hepatic and peripheral insulin resistance, hyperglycemia, and dyslipidemia.
4) The metabolic “signature” of chronic stress
What changes in glucose, fat, and protein pathways
Below is a general pattern—not a diagnosis, but a mechanistic map.
4.1 Glucose: from “fuel availability” to insulin resistance
Under chronic activation, glucocorticoids can:
- increase hepatic glucose production (gluconeogenesis)
- impair insulin signaling in tissues (skeletal muscle, liver, adipose), contributing to insulin resistance
This helps explain why chronic stress physiology often overlaps with pathways involved in metabolic syndrome and type 2 diabetes risk (again: risk pathways, not a guarantee).
4.2 Lipids: mobilization → ectopic fat → signaling disruption
Stress-driven sympathetic activity increases lipolysis, raising circulating free fatty acids.
Glucocorticoids can further promote lipid redistribution and lipid accumulation in liver and muscle—changes associated with insulin resistance.
From a chemistry standpoint, elevated fatty acids can feed into lipid intermediates (e.g., ceramides in some models) that interfere with insulin signaling—one reason glucocorticoid-induced metabolic disruption is studied at the level of lipid biochemistry.
4.3 Protein: “emergency substrate” costs
In prolonged stress, the body may increase protein breakdown to provide amino acids for gluconeogenesis and acute needs. This is adaptive during emergency, but persistent catabolic signaling can impair repair, muscle maintenance, and metabolic stability.
5) Stress and inflammation: when the brake stops working
A central reason chronic stress can contribute to chronic disease is that it may push the body into a state of inflammatory bias.
5.1 The paradox: cortisol is anti-inflammatory, yet chronic stress can increase inflammation
One influential model proposes that chronic stress can produce glucocorticoid receptor resistance, meaning cortisol signaling becomes less effective at shutting down inflammatory responses.
When GR signaling is impaired, pro-inflammatory transcription programs can become disinhibited.
5.2 NF‑κB as a molecular “switchboard” for inflammation
NF‑κB is a pivotal transcription factor that regulates many inflammatory genes (cytokines, chemokines, immune activation programs).
Reviews describe reciprocal interference between GR signaling and NF‑κB signaling—helping explain how chronic stress states can shift the balance toward sustained inflammatory activity.
5.3 Why this matters for many chronic diseases
Persistent low-grade inflammation is a common pathway implicated across:
- cardiometabolic disease
- some autoimmune conditions
- mood and neuroinflammatory disorders
- aspects of tumor microenvironment biology (more on this carefully below)
A 2024 immunology review summarizes that stress can alter immune cell distribution/trafficking and modulate immune function, affecting susceptibility and health.
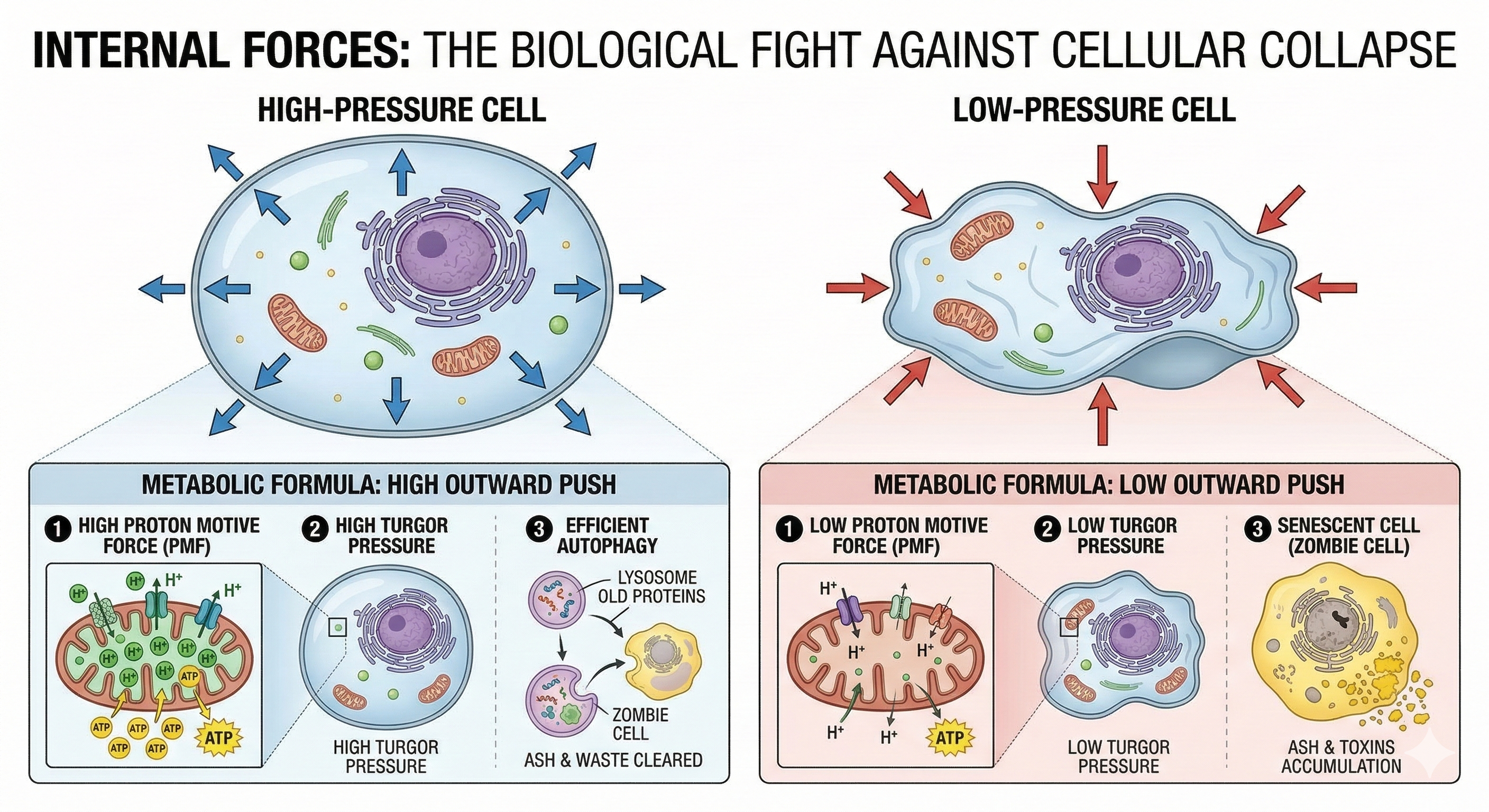
6) Mitochondria: the cellular bridge between “stress signals” and chronic wear
If you want one concept that unites chemistry and physics in stress biology, it’s mitochondria.
Mitochondria convert nutrients into ATP, regulate reactive oxygen species (ROS), shape inflammatory signaling, and influence cell survival programs. The “energetic view of stress” literature emphasizes reciprocal links: stress hormones influence mitochondrial function and energy substrate distribution, and mitochondrial state can shape stress reactivity.
This helps connect chronic stress to:
- fatigue states
- metabolic dysregulation
- inflammatory amplification
- accelerated biological aging pathways
SDAE translation: when energy production and energy allocation become chronically distorted, coherence collapses at the cellular level before symptoms fully surface.
7) Stress, aging, and “time”: telomeres as one window
A deep SDAE perspective, and here it fits: stress can change the body’s relationship with time.
Telomeres (protective chromosome-end structures) are often used as one biomarker of cellular aging processes. A systematic review and meta-analysis found an association between perceived psychological stress and shorter telomere length (with important methodological caveats and heterogeneity across studies).
A longitudinal study also reported that increases in chronic stress over time were associated with telomere shortening over a decade.
SDAE translation: chronic stress doesn’t only hurt “today.” It can reshape the body’s long-term maintenance signals—how it spends repair resources across time.
8) Chronic disease progression in general terms: a unifying pathway
Across many chronic diseases, a recurring pattern appears:
- Repeated stress signaling (SNS/HPA activation)
- Metabolic rerouting (glucose up, lipolysis up, altered insulin signaling)
- Immune modulation (cell trafficking shifts, inflammatory programs skew)
- Mitochondrial and oxidative strain (energy inefficiency + inflammatory reinforcement)
- System-level outcomes: insulin resistance, vascular strain, sleep disruption, mood changes, flare vulnerability, slower repair
This is one reason “stress management” isn’t just psychological comfort—it can be a legitimate part of a broader health-support strategy alongside medical care. A 2024 meta-analysis of stress management interventions found overall beneficial effects on cortisol outcomes across randomized trials (with variability depending on measurement and intervention type).
9) A careful note about cancer (because people ask)
It is scientifically reasonable to say that chronic stress biology can influence immunity and signaling pathways that may affect aspects of tumor microenvironments and progression in some contexts—but it is not scientifically responsible to claim “stress causes cancer” as a simple statement.
Recent reviews summarize mechanisms by which stress mediators can affect immune surveillance, inflammation, and tumor biology, while emphasizing complexity and variability.
SDAE framing: stress is best understood as a risk amplifier and a resilience reducer—not a single cause. Stress doesn’t cause mutation. Stress biology can lead to mutation.
10) The SDAE integration: knowledge → coherence → action
SDAE knowledge drives more concrete action.
SDAE would describe this as:
- Knowledge restores agency (Inner Reality regains authorship).
- Agency enables repetition (practice becomes consistent).
- Consistency builds coherence (biology responds to stable inputs over time).
- Coherence reduces warping (your “spacetime” metaphor becomes physiological: steadier rhythms, fewer spikes, more recovery).
That’s why education is not “extra.” In SDAE, education is an intervention on the Inner Reality that changes the Outer Reality choices—sleep timing, diet structure, movement, boundaries, community support, clinical follow-through.
11) A simple teaching model I use with NuSpecians
Here’s a non-medical, science-accurate framing that people remember:
Stress → Signals → Substrates → Structure → Symptoms
- Signals: cortisol, catecholamines
- Substrates: glucose, fatty acids, amino acids
- Structure: mitochondria, immune tone, vascular tone
- Symptoms: fatigue, dysregulation, flares, metabolic drift, mood strain
SDAE add-on line: the Astonian signature, applied scientifically, for the whole family:
“What bends your time… bends your biology.”

